
This enables ice to be lighter than water, allowing it to float. Hence, its ρ water decreases and becomes more than the initial volume.

It continues to contract and shrink in volume as it cools until it reaches 4☌.īeyond this temperature, its volume expands and exceeds the starting volume. Due to this molecular structure, water behaves abnormally when cooled. To understand the concept of ice floating on water, we must first study the structure of water.Įvery water molecule is made up of two hydrogen atoms bonded to one oxygen atom. Check the full article “Melting point of water”. However, this exceptional behavior is never seen in nature for any of these materials except water because melting points are very high except for water. Some materials like Silicon, germanium, bismuth, and water are less dense in solid form than in liquid form. Therefore, the ρ solid > ρ liquid > ρ gas Solid atoms or molecules are more closely packed together than liquid atoms or molecules. In general, a solid mass of a substance sinks when immersed in a liquid of the same substance. The less-dense stuff floats on top of the dense liquid.īecause the ρwater of ice is smaller than the ρwater of water, ice floats on water. The density of water (g/ml) = 8kg/(8 x10-3 m3 ) The mass (m) of 8 liters of water is 8 kg. Specific weight (γ) = (ρ x volume x g)/volume It is represented by the symbol γ.Īt 4☌, the specific weight of water on earth is 9.807 kN/m 3 = 62.43 lbs/ft 3.Ĭheck the full article “Specific gravity of water”. The specific weight of water is the weight of water per unit volume. There are three different Units of Gallon: Weight of a Gallon of Water Weight of 1 liter (l) of pure water (at 4 ☌) = 1 kilogram (kg). The weight of a gallon of water depends upon storing the temperature of the water, For the sake of the question, ‘How much does a gallon of water weigh?’, we are going to focus on the weight of water at 62 ☏ (17 ☌).ġ US gal of water = 8.345 pounds = 3.785 kg (at 17 ☌).ġ Imperial gal of water = 10.02 pounds = 4.545 kg (at 17 ☌).

At 4☌, the density of fresh water on Earth is generally understood to be 1000 kg/m 3.
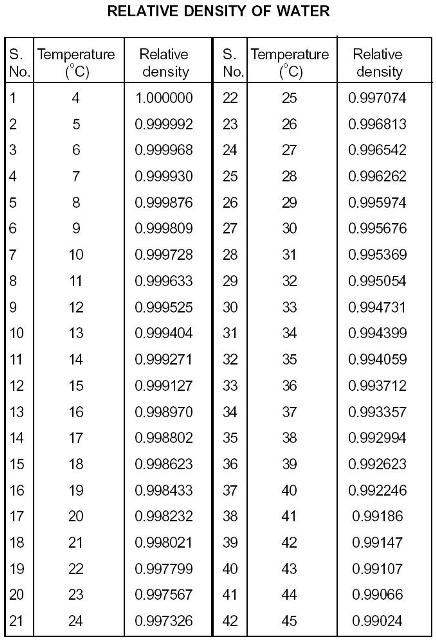
However, this value varies depending on the temperature and impurities in the water.Ĭommon units of ρ water are grams per milliliter (1 g/ml), grams per cubic centimeter ( g/cm 3), and pounds per cubic foot (lb/ft 3). The density of water (ρ water) is around 8.345 lbs/U.S. This is since iron has a greater amount of substance than wood.ĭensity (ρ) formula = mass (m) of substance ÷ volume (v) of substance The density of water in lbs/U.S. We’ll find that iron is heavier than wood. It is the mass-to-volume ratio of the material. How much does a gallon of water weigh at its highest density?ĭensity refers to the amount of substance per unit volume.The density of Water in g/ml (Effect of Temperature).Calculating the specific weight of water.Last accessed: 29 August 2020 (.gov/compound). National Library of Medicine 8600 Rockville Pike Bethesda, MD 20894 USA. National Center for Biotechnology Information U.S. Department of Commerce 100 Bureau Drive Gaithersburg, MD 20899 USA. National Institute of Standards and Technology U.S. Glucose density values in 285 units of density, in the form of a matrix Density = weight ÷ volume Glucose density values, grouped by weight and shown as value of density, unit of density grain per. See density of Glucose in hundreds of units of density measurement grouped by weight.Density of Glucose lb ft3 = 97.39 lb/ft³.Density of Glucose lb in3 = 0.056 lb/in³.Density of Glucose g mm3 = 0.0016 g/mm³.Density of Glucose in a few select units of density measurement:.Melting Point (MP), Glucose changes its state from solid to liquid at 146☌ (294.8☏ or 419.15K)Įlements: Carbon (C), Hydrogen (H), Oxygen (O).In Imperial or US customary measurement system, the density is equal to 97.388 pound per cubic foot, or 0.9017 ounce per cubic inch. density of glucose is equal to 1 560 kg/m³. Glucose weighs 1.56 gram per cubic centimeter or 1 560 kilogram per cubic meter, i.e.
